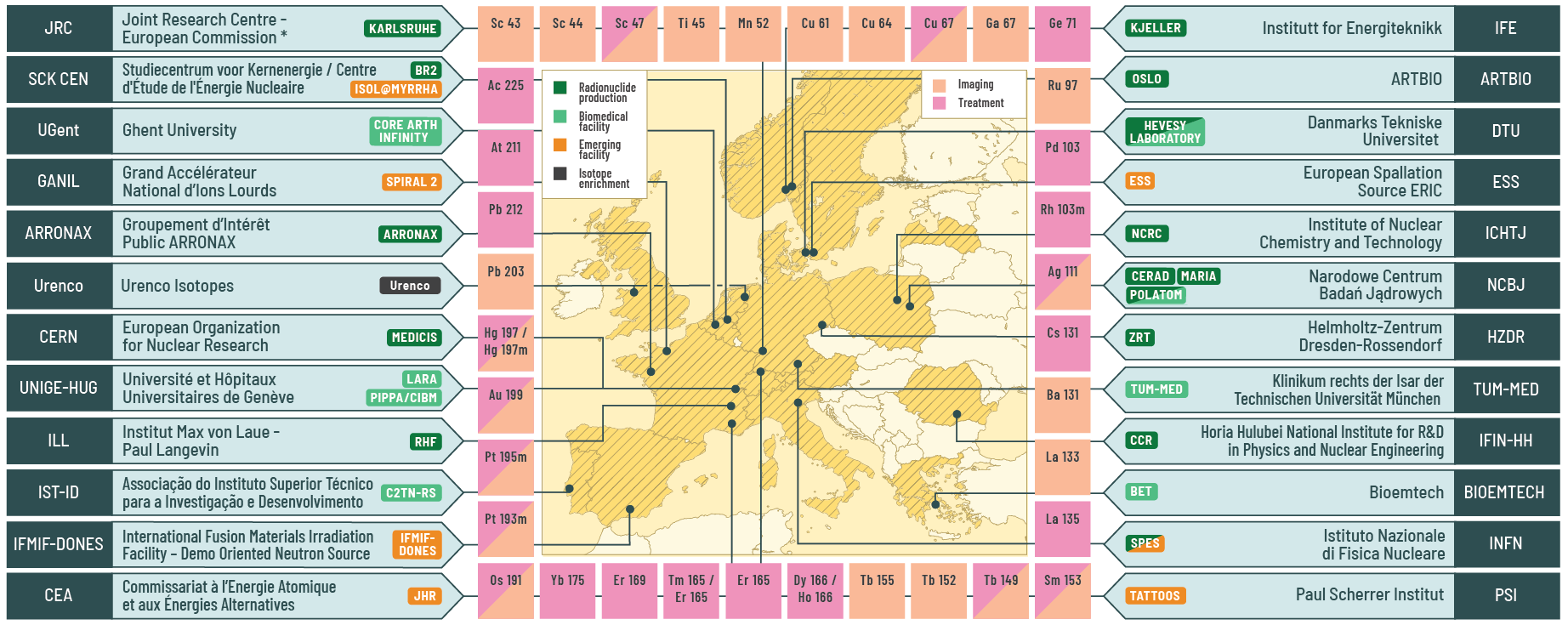
PRISMAP final press release
To mark the completion of the PRISMAP project, we have just published a press release providing an overview of the user projects funded by our European programme for medical radionuclides. The press release includes a delivery map where you can track all radionuclide deliveries made during the project's lifetime.
Enjoy reading!

Videos and presentation from Warsaw now available
We are happy to announce that the videos and presentations from our final public event in Warsaw are now available on its dedicated page! Scroll through the agenda, and you will find the related information for each presentation on the right.
PRISMAP Final public event 18-19 November, Warsaw and Online
Join the final public event of PRISMAP – The European medical radionuclides programme on 18-19 November 2025 at the Heavy Ion Laboratory (HIL), University of Warsaw, Ludwika Pasteura 5A, with a hybrid participation option. The meeting is hosted by the National Centre for Nuclear Research Radioisotope, Centre POLATOM, a PRISMAP partner.
We will showcase highlights from PRISMAP user projects, key lessons from building this research infrastructure, and perspectives. The agenda features talks by experts from CERN, SCK CEN, INFN, ILL, TRIUMF and MSKCC; as well as sessions on regulatory requirements and industry collaboration, plus dedicated networking.
Registration & posters
Participation is free but requires registration via the event page. Please register by 7 November 2025. All participants are invited to bring a poster to the poster session – an excellent opportunity to present your research and connect with the European radiopharmaceutical community. We particularly encourage early-career researchers to come and present their work!
Zapraszamy do udziału w finałowym wydarzeniu projektu PRISMAP – The European medical radionuclides programme, które odbędzie się 18-19 listopada 2025 r. w Środowiskowym Laboratorium Ciężkich Jonów (ŚLCJ), Uniwersytet Warszawski, ul. Ludwika Pasteura 5A, Warszawa, z możliwością udziału online. Gospodarzem spotkania jest Narodowe Centrum Badań Jądrowych Ośrodek Radioizotopów POLATOM – partner konsorcjum PRISMAP.
Podczas wydarzenia podsumujemy najważniejsze osiągnięcia projektów użytkowników PRISMAP, wnioski z czteroletniej pracy nad infrastrukturą badawczą oraz kierunki dalszego rozwoju. W programie m.in. prezentacje przedstawicieli CERN, SCK CEN, INFN, ILL, TRIUMF, MSKCC oraz sesja dotycząca wymogów regulacyjnych i współpracy z przemysłem.
Zgłoszenia i plakaty
Udział jest bezpłatny, lecz wymaga rejestracji poprzez formularz dostępny na stronie wydarzenia. Prosimy o rejestrację do 7 listopada 2025 r. Zachęcamy do przywiezienia plakatu i udziału w sesji plakatowej – to świetna okazja do prezentacji wyników badań i nawiązania współpracy w europejskiej społeczności radiofarmaceutycznej. Szczególnie zachęcamy badaczki i badaczy na wczesnym etapie kariery do udziału i prezentacji swoich prac!
PRISMAP Webinar "from PRISMAP to PRISMAP+", recording now available
We are pleased to announce that the recording of our webinar "From PRISMAP to PRISMAP+" is now available on its dedicated page. Enjoy watching!
Join the CAS course on Medical Accelerators
In collaboration with Riga Technical University, the CERN Accelerator School is proud to present a CAS course on Medical Accelerators.
Particle accelerators, known since decades as cornerstones of fundamental physics research, nowadays evolved into indispensable tools for society across diverse fields. This course focuses on profound and widespread accelerator applications in medicine, particularly in oncology and in nuclear medicine. By bridging the communities of accelerator science and medical physics, we will provide a comprehensive overview of the field from both technological and clinical perspectives.
For more information and registration, please visit the dedicated webpage of the course.
PRISMAP back from EANM
PRISMAP has just returned from the annual congress of the European Association of Nuclear Medicine (EANM – Barcelona, 6–8 October 2025). With 9,431 congress participants (including online participants) and 2,991 abstracts submitted, this year's congress set a new record. On the occasion of the 40th anniversary of the EANM Congress, PRISMAP and its representatives were more present than ever, underlining the growing importance of PRISMAP.
The highlight of PRISMAP's contributions to the congress was a dedicated session on Sunday, 5 October 2025. As part of a thematic block on EU projects, PRISMAP was able to share its results and perspectives. To familiarise the audience with PRISMAP, Thierry Stora from CERN first presented the structure and scope of PRISMAP, the number of projects funded and the development prospects. Eleni Gourni, Bern, Switzerland, reported on her experiences as a PRISMAP user with Sc-47 from PRISMAP. She described the process from applying for a PRISMAP user project to its successful implementation. Finally, Clemens Decristoforo from Innsbruck, Austria, explained how PRISMAP was involved in networking activities with a focus on harmonisation and standardisation. The session ended with a lively discussion among interested users about logistics and supply chains, the application process and other questions about the scope and future of the project, underscoring the great interest and need for novel radionuclides for medical applications.
PRISMAP plays an important role in current political decision-making processes at European Commission level, as was also made clear at the EANM Congress. According to Paola Erba, President of EANM, and Wim Oyen (former President and Board Member of EANM), PRISMAP acts as an EU platform for research in the field of biomedical radionuclides and is in contact with various institutions, in particular the European Medicines Agency.
In addition, PRISMAP was represented by a number of speakers, poster presenters and chairs from the consortium, as well as by users who presented their projects made possible by PRISMAP's services. We are particularly pleased that three of our presentations were classified as ‘top rated oral presentations’ by the EANM:
- N. Kumar, E. Gourni et al: Targeted Radionuclide Therapy of Prostate Cancer using a177 Lu- and47 Sc-labeled Gastrin-Releasing Peptide Receptor Antagonist. Evaluation as Monotherapy and in Combination with Everolimus
- A. Gustafsson, M. Nestor et al: Tb, or Not Tb - Exploring Terbium-161 for CD44v6Targeted Radionuclide Therapy
- M. Salek, J. W. Engle et al: Small cyclotron production of n.c.a.103 Pd from Rh targets for targeted radionuclide therapy

Webinar "from PRISMAP to PRISMAP+"
PRISMAP will soon come to an end, with a closing date of 31 December 2025.
On Tuesday, 14 October 2025 from 16.30 to 17.15 CEST, PRISMAP coordinator Thierry Stora will present our view of PRISMAP's achievements and future prospects in a follow-up project in a 45-minute webinar, including a short presentation followed by an open discussion.
For more information and the meeting link please refer to our dedicated event page.
PRISMAP @ EANM 2025
Below is an overview of PRISMAP members’ participation at the 2025 EANM conference, including poster sessions, presentations, and details of the special PRISMAP Session on Sunday, 5 October.
Join our special session: a unique opportunity to learn about PRISMAP’s journey, user experiences, and translational applications of its radionuclides in nuclear medicine research and therapy.
5 October 14:00—14:30 Room 121
- Thierry Stora – The PRISMAP project Past & Future (10 min)
- Eleni Gourni – A PRISMAP user’s experience (5 min)
- Clemens Decristoforo – PRISMAP radionuclides – translational activities (5 min)

5 October 8:32—8:36 Poster presentation
Dr. Naveen Kumar — Preclinical Evaluation of a 47Sc-labeled Gastrin Releasing Peptide-based Radioantagonist for the Targeted Management of Breast Cancer

5 October 9:16—9:20 Poster presentation
Melissa Crabbé — The absorbed dose-response relationship of the Auger electrons from La-135

7 October 8:30—8:40 Poster presentation
Dr. Naveen Kumar — Targeted Radionuclide Therapy of Prostate Cancer using a 177Lu- and 47Sc-labeled Gastrin-Releasing Peptide Receptor Antagonist. Evaluation as Monotherapy and in Combination with Everolimus

Poster presentation
Kristof Baete — Nuclear medicine equipment standardisation for clinical translation: a case study with Tb-161

Poster presentation
D. Pawlak and R. Mikołajczak — Small-scall production of 161Tb
From PRISMAP to PRISMAP+
PRISMAP will soon come to an end, with a closing date of 31 December 2025.
This opens the door towards the next step: We have just submitted a proposal for a successor project PRISMAP+, which we hope the EC will select for funding so that we can start it in late Spring 2026.
Our help desk will remain open during the transition phase, and interested users are invited to contact us through this channel.
A first call for user projects under PRISMAP+ would be opened towards the end of 2026.
On Tuesday, 14 October 2025, PRISMAP coordinator Thierry Stora will present our view of PRISMAP's achievements and future prospects in a follow-up project in a 45-minute webinar, including a short presentation followed by an open discussion.
To get the full picture and celebrate with us, join us at the PRISMAP closing conference, which will take place in Warsaw and online on 18 and 19 November 2025. There, we will present the final results of PRISMAP and our plans for the future in detail.
To keep informed, make sure you are registered as part of the PRISMAP community.
Save the date: PRISMAP fInal event
The PRISMAP Final Event will take place in Warsaw this coming fall. More information and details will be available on its dedicated page. In the meantime, save the date — 19 November — and stay tuned for further updates!
PRISMAP's consortium meeting 8
Our workshop on emerging infrastructures was immediately followed by our 8th consortium meeting: another 2.5 days were dedicated to the lessons learnt from the project so far and the plans for the final project phase and beyond. In addition, we had the pleasure of meeting Belgian companies active in the field of medical radionuclides during a ‘PRISMAP meets industries’ session. Our public event, which has already become a tradition at our consortium meetings, rounded off the meeting with our project news and exciting reports from our user projects. Many, many thanks to SCK-CEN, our host in this beautiful lake district near Mol/Belgium!

PRISMAP workshop on emerging infrastructures, 31 Mar-2 April 2025, Mol/BE
We have just returned from the second PRISMAP workshop on emerging infrastructures - two intensive days of presentations, poster exhibition and discussions, as well as a half-day visit to the SCK CEN infrastructures MYRRHA, BR2 and CRF.
The programme included sessions on the assessment of emerging infrastructures for specific radionuclides, isotope separation for isotope enrichment and technical developments for radionuclide production. A round table discussion on innovative radionuclides and user needs rounded off the workshop.
Stay tuned, a white paper will be produced based on this workshop

First mass-separated ion beam at SCK CEN
Researchers at our partner organisation SCK CEN have recently achieved a new milestone in the development of ISOL@MYRRHA: the first production and mass separation of an ion beam. The team successfully separated the stable isotopes of rubidium using a newly developed ISOL system installed for off-line operation at SCK CEN. More information you can find on the SCK CEN website.
PRISMAP session at Norwegian workshop - 12-16 May 2025, Oslo
A PRISMAP session will take place at the Workshop on radionuclide production and medical application (Oslo, 12-16 May 2025), organised by the new Norwegian Nuclear Research Centre at University of Oslo and including a discussion on the preparation of the PRISMAP+ successor project and a Scandinavian node.
You are all very welcome to join and submit an abstract, preferentially by 1 Mar 2025.
Some grants may be made available to young researchers for travel costs.
Stay tuned, more information and registration will be made available soon.
Public event related to Riga school - 21 Feb 2025, 10-12h30 CET
You are all cordially invited to the public event, which is being organised in parallel with the upcoming PRISMAP school "Current and Innovative Radionuclides for Medical Imaging and Therapy" in Riga.
The event aims to highlight advancements in the multidisciplinary field of nuclear medicine, including radiopharmaceutical sciences, radioisotope production, clinical applications, and technological solutions.It is designed for medical doctors, residents, trainees, young researchers, and students at the Bachelor, Master, or PhD level in nuclear medicine, physics, or chemistry, as well as anyone with an interest in the field.
For more details regarding the programme and registration, please refer to the dedicated page on the PRISMAP website.